B·R·A·H·M·S MR-proADM
Reliable measurement of ADM is complicated by a number of issues, such as a short half-life, a fast metabolism, low concentrations, a rapid degradation by proteases, and binding to compliment factor H. Accordingly, ADM levels can therefore be typically underestimated. The measurement of mid-regional proadrenomedullin (MR-proADM) provides a solution to these problems.
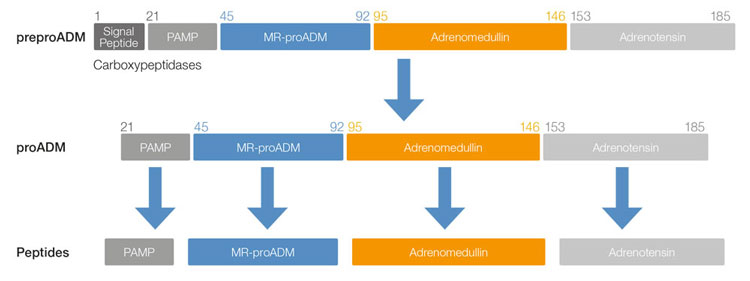
Biosynthesis of MR-proADM
MR-proADM is a fragment of 48 amino acids which splits from proADM molecule in a 1:1 ratio with Adrenomedullin, and therefore proportionally represents the levels and activity of Adrenomedullin. Its biological inactivity means that it is not involved in the binding to vessel walls and surfaces commonly found with Adrenomedullin, and its longer half-life of several hours and biologically inactivity results in a more accurate estimation of plasma concentration levels than using ADM. Ref-1,2
B·R·A·H·M·S MR-proADM KRYPTOR
The measurement of MR-proADM concentrations provides a more precise assessment of disease severity and patient risk management, and can enhance clinical investigation and treatment decisions. Ref-3-7 The rapid triaging and risk assessment of patients upon emergency department presentation can decrease time to treatment, increase out-patient numbers and reduce length of hospitalisation. Ref-8 In the intensive care department, it can provide an immediately assessment of disease severity in order to maximise patient safety, guide the most appropriate treatment and provide an early warning of developing complications. Ref-9
As described in the instruction for use, the B·R·A·H·M·S MR-proADM KRYPTOR assay is indicated for use as an aid together with other clinical evaluation and laboratory diagnostic findings to assess outcome and risk in patients with conditions such as lower respiratory tract infections, urinary tract infections and kidney disease, as well as in patients suffering from sepsis, severe sepsis and septic shock. It is also of use in risk assessment in the Emergency Department setting.
MR-proADM levels are not influenced by food or water intake, with no significant gender related differences. The biomarker is extremely stable, with no degradation observed up to 24 hours at room temperature. Ref-10
 Levels are not influenced by food or water intake
Levels are not influenced by food or water intake
 No significant gender related differences
No significant gender related differences
 Stability of up to 24 hours in EDTA plasma at room temperature and over four freeze / thaw cycles
Stability of up to 24 hours in EDTA plasma at room temperature and over four freeze / thaw cycles
 Well documented for clinical use
Well documented for clinical use
 Rapidly available to aid timely clinical decision-making using the KRYPTOR platform*
Rapidly available to aid timely clinical decision-making using the KRYPTOR platform*
* assay incubation time 29 mins and small sample volume (26 μL in EDTA plasma)
References B·R·A·H·M·S MR-proADM KRYPTOR
Ref-1: Struck et al. 2004; Peptides (25 (8): 1369-72.
Ref-2: Caruhel et al. 2009; Clin Biochem 42 (7-8):725-8.
Ref-3: Renaud et al. 2012; Chest 142 (6): 1447-1454.
Ref-4: Saeed et al. 2019; Crit Care 23 (1): 40.
Ref-5: Albrich et al. 2011; BMC Infect Dis 11 (1): 112.
Ref-6: Citgez et al. 2018; Chest 154 (1): 51-57
Ref-7: Brusse-Keizer et al. 2015; Respir Med 109 (6): 734-42.
Ref-8: Gonzalez del Castillo et al. 2021; European Journal of Internal Medicine 88: 104-113.
Ref-9: Elke et al. 2018; Crit Care 22 (1): 79.
Ref-10: Morgenthaler et al 2005; Clin Chem 51(10: 1823-1820.